Time:2025-11-17

Eicosapentaenoic Acid Ethyl Ester (EPA-EE), also known as EPA ethyl ester, is an Omega-3 fatty acid derivative derived from eicosapentaenoic acid (EPA). In 2012, Vascepa (brand name) developed by Amarin was approved by the U.S. FDA as a prescription medication to reduce the risk of atherosclerotic diseases. It has gained significant market presence globally, with its clinical benefits widely recognized.
Domestic drug review databases reveal that since Chengdu Guowei Pharmaceutical Group filed a Class 3 generic drug application, nearly 40 companies have subsequently applied for generic versions. Approved manufacturers include Sichuan Guowei, Yangtze River Pharmaceutical, Renfu Puke, Kelun Pharmaceutical, Xinhua Pharmaceutical, Hengrui Medicine, and Qilu Pharmaceutical. Our internal sales data indicates that, including manufacturers yet to apply, the number of companies developing this product is conservatively estimated at no fewer than 60, making it a highly sought-after generic drug candidate.

I. Research Background
The prevalence of severe hypertriglyceridemia (TG) in China is approximately 1.4%, with a treatment rate of 6.84%, and about 1.09 million patients with severe hyperTG are receiving treatment. In addition to dietary control, this product is used to reduce TG levels in adult patients with severe hypertriglyceridemia (≥500mg/dL). The drug has a dual lipid-lowering mechanism, assisting in the management of hyperTG and the prevention and control of cardiovascular diseases. It also has a unique metabolic pathway, meeting the needs of patients with hepatic or renal insufficiency and those intolerant to statins for TG reduction. The current main sales channel for ethyl eicosapentaenoate soft capsules is retail pharmacies (physical pharmacies in cities + online pharmacies), accounting for over 90%. The product's sales in China's retail pharmacies exceeded 12 million yuan in 2024, with a growth rate of 421.46% in Q12025, and its market potential is continuously being unleashed.
Due to the sensitivity of ethyl eicosapentaenoate to strong acids, strong bases, high temperature, and strong light, the cis-dual bond in its structure may undergo isomerization, resulting in cis-trans isomer impurities. To ensure strict quality control of ethyl eicosapentaenoate in pharmaceuticals, our company has conducted research on 16 related substances of ethyl eicosapentaenoate.

Figure 1: Structure of ethyl eicosapentaenoate and related substances
II. Research on HPLC Method
After performing NMR and MS qualitative analysis on the impurities of ethyl eicosapentaenoate, our company conducted gas chromatography analysis according to the impurity method specified in the registration standard for ethyl eicosapentaenoate soft capsules. The results are shown in Figures 2 and 4 below.

Figure 2: Liquid chromatogram of ethyl eicosapentaenoate and related substances
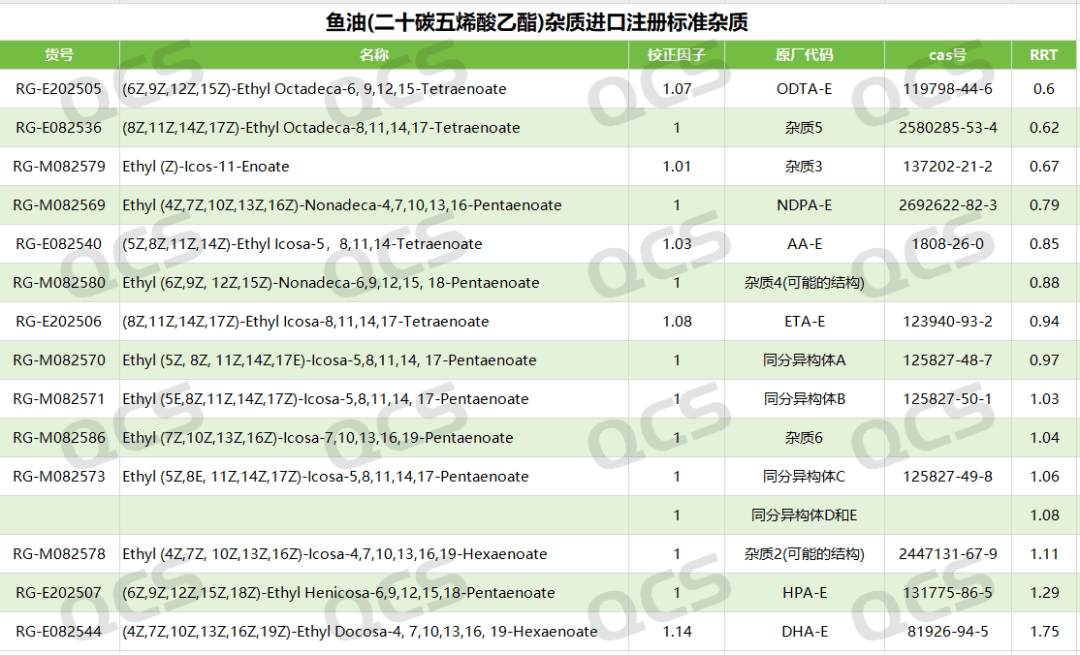
Figure 3: Information on related substances in the registration standards for ethyl docosahexaenoate soft capsules
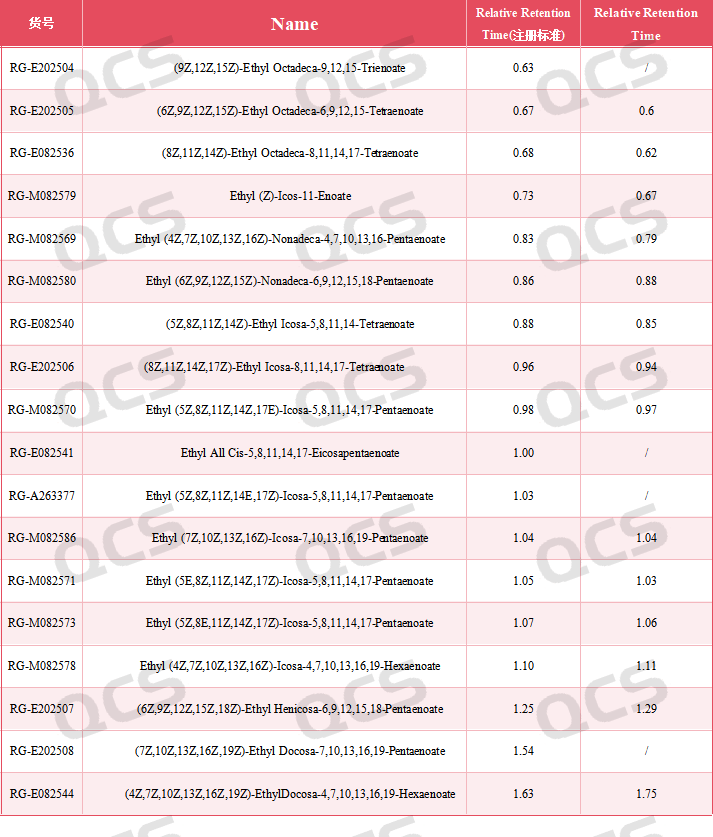
Figure 4: Relative retention time data of ethyl eicosapentaenoate related substances
The results show that the relative retention time of all the impurities is within the standard requirement.
3. Summary
Through standardized methods, our company maintains strict quality control over ethyl eicosapentaenoate impurities. As one of our top-selling impurity series, this product line features complete chromatographic profiles, high purity, competitive pricing, and customization options. For more information about related products and services, please feel free to inquire.



Eicosapentaenoic Acid Ethyl Ester (EPA-EE), also known as EPA ethyl ester, is an Omega-3 fatty acid derivative derived from eicosapentaenoic acid (EPA). In 2012, Vascepa (brand name) developed by Amarin was approved by the U.S. FDA as a prescription medication to reduce the risk of atherosclerotic diseases. It has gained significant market presence globally, with its clinical benefits widely recognized.
Domestic drug review databases reveal that since Chengdu Guowei Pharmaceutical Group filed a Class 3 generic drug application, nearly 40 companies have subsequently applied for generic versions. Approved manufacturers include Sichuan Guowei, Yangtze River Pharmaceutical, Renfu Puke, Kelun Pharmaceutical, Xinhua Pharmaceutical, Hengrui Medicine, and Qilu Pharmaceutical. Our internal sales data indicates that, including manufacturers yet to apply, the number of companies developing this product is conservatively estimated at no fewer than 60, making it a highly sought-after generic drug candidate.

I. Research Background
The prevalence of severe hypertriglyceridemia (TG) in China is approximately 1.4%, with a treatment rate of 6.84%, and about 1.09 million patients with severe hyperTG are receiving treatment. In addition to dietary control, this product is used to reduce TG levels in adult patients with severe hypertriglyceridemia (≥500mg/dL). The drug has a dual lipid-lowering mechanism, assisting in the management of hyperTG and the prevention and control of cardiovascular diseases. It also has a unique metabolic pathway, meeting the needs of patients with hepatic or renal insufficiency and those intolerant to statins for TG reduction. The current main sales channel for ethyl eicosapentaenoate soft capsules is retail pharmacies (physical pharmacies in cities + online pharmacies), accounting for over 90%. The product's sales in China's retail pharmacies exceeded 12 million yuan in 2024, with a growth rate of 421.46% in Q12025, and its market potential is continuously being unleashed.
Due to the sensitivity of ethyl eicosapentaenoate to strong acids, strong bases, high temperature, and strong light, the cis-dual bond in its structure may undergo isomerization, resulting in cis-trans isomer impurities. To ensure strict quality control of ethyl eicosapentaenoate in pharmaceuticals, our company has conducted research on 16 related substances of ethyl eicosapentaenoate.

Figure 1: Structure of ethyl eicosapentaenoate and related substances
II. Research on HPLC Method
After performing NMR and MS qualitative analysis on the impurities of ethyl eicosapentaenoate, our company conducted gas chromatography analysis according to the impurity method specified in the registration standard for ethyl eicosapentaenoate soft capsules. The results are shown in Figures 2 and 4 below.

Figure 2: Liquid chromatogram of ethyl eicosapentaenoate and related substances

Figure 3: Information on related substances in the registration standards for ethyl docosahexaenoate soft capsules

Figure 4: Relative retention time data of ethyl eicosapentaenoate related substances
The results show that the relative retention time of all the impurities is within the standard requirement.
3. Summary
Through standardized methods, our company maintains strict quality control over ethyl eicosapentaenoate impurities. As one of our top-selling impurity series, this product line features complete chromatographic profiles, high purity, competitive pricing, and customization options. For more information about related products and services, please feel free to inquire.


Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*All our products are for R&D.

*All our products are for R&D.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号